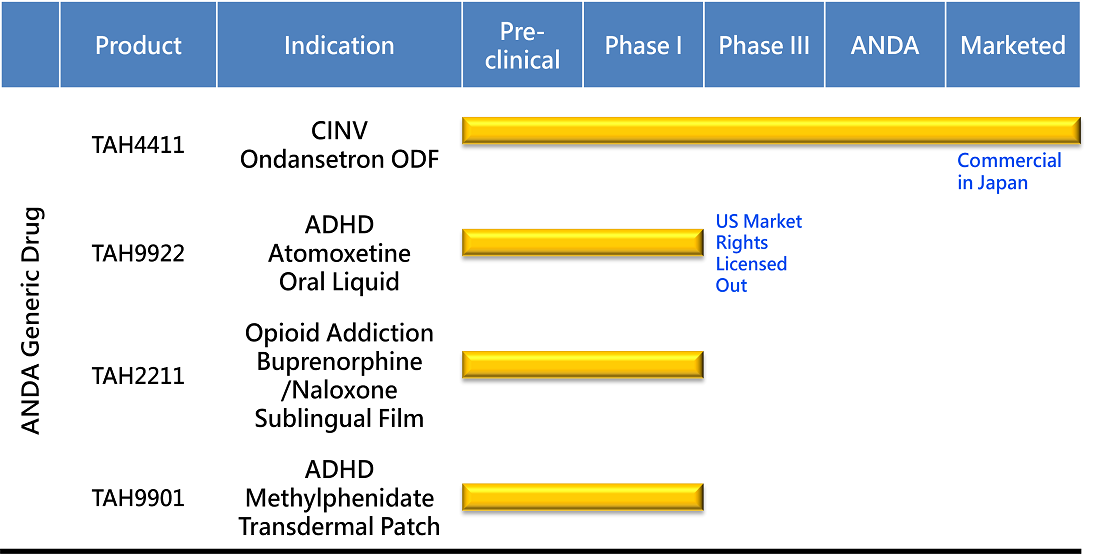
PIPELINE
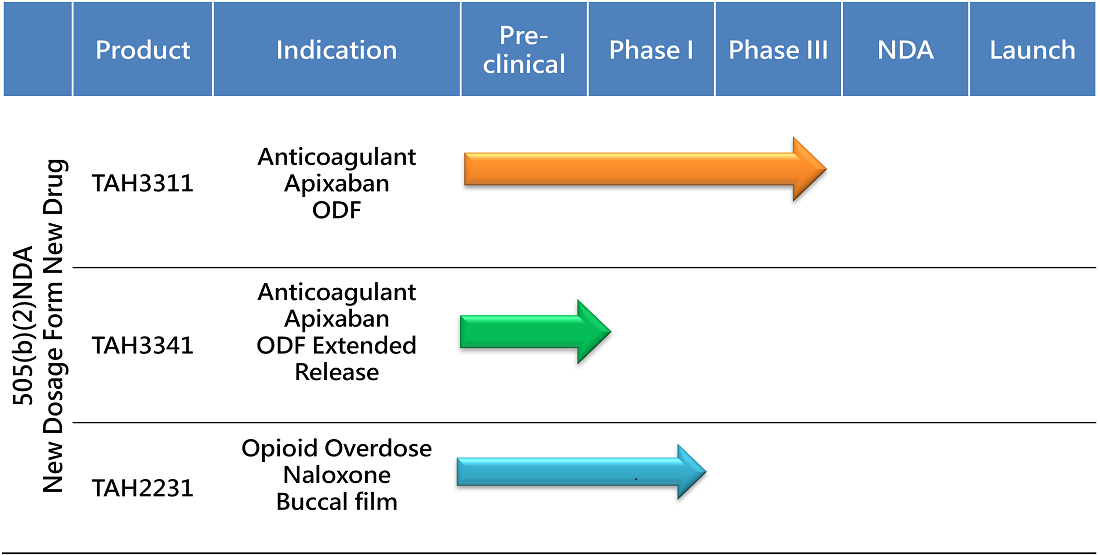
The 505(b)(2) NDA pathway streamlines drug development by leveragingexisting safety and efficacy data, reducing timelines, costs, and risks comparedto traditional methods. This approach accelerates market entry, offeringpatients safe, effective, and innovative treatments while addressing unmetneeds like improved dosage forms and tailored solutions for specificpopulations. TAHO leverages this pathway to deliver better therapies, bridgeinnovation with accessibility, and drive progress in healthcare.